Y90 Radioembolization
If you or a loved one has been diagnosed with colorectal cancer that has spread (metastasized) to the liver, it is important to learn the facts and evaluate treatment options. Of the nearly 140,000 Americans diagnosed with colorectal cancer every year, at least 60 percent will see their cancer spread to the liver, and many will die of this disease.1, 2 A diagnosis of colorectal liver metastases can be overwhelming, but there is hope.
 SIR-Spheres® microspheres treatment, offered at Memphis Clinic, is a targeted radiation therapy that delivers a dose of internal radiation up to 40 times higher than conventional external beam therapy while sparing healthy tissue. This maximizes the treatment’s effectiveness and reduces the risk of injury to the liver. The treatment is typically administered as an outpatient procedure at a local treating hospital. SIR-Spheres microspheres are FDA PMA approved for patients with inoperable metastatic colorectal cancer to the liver.*
SIR-Spheres® microspheres treatment, offered at Memphis Clinic, is a targeted radiation therapy that delivers a dose of internal radiation up to 40 times higher than conventional external beam therapy while sparing healthy tissue. This maximizes the treatment’s effectiveness and reduces the risk of injury to the liver. The treatment is typically administered as an outpatient procedure at a local treating hospital. SIR-Spheres microspheres are FDA PMA approved for patients with inoperable metastatic colorectal cancer to the liver.*
Who is a candidate for the treatment?
This treatment is indicated for patients with colorectal liver cancer whose liver metastases cannot be surgically removed. As with any treatment, the medical team at Memphis Clinic can help you decide if this treatment is best suited for your condition.
 How are SIR-Spheres microspheres administered?
How are SIR-Spheres microspheres administered?
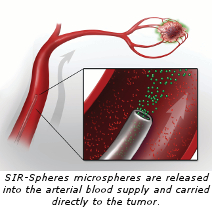
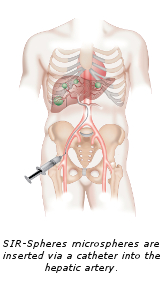
SIR-Spheres microspheres are administered through a process called Selective Internal Radiation Therapy (SIRT). During the procedure, an interventional radiologist makes a small puncture, usually into the femoral artery near the groin. A small flexible tube, known as a catheter is then guided through the artery into the liver and millions of microscopic spheres (about a third of the width of a human hair), are delivered directly into the tumor. The entire procedure takes about 90 minutes. Patients will be sleepy during the procedure but able to communicate with the doctor and the team. Most patients return home four to six hours following treatment.
How do SIR-Spheres microspheres work?
The SIRT procedure delivers radiation, which is often used to treat cancer, directly into the liver tumors by using the tumor’s own blood supply. Normal liver tissue takes about 90 percent of its blood supply from the veins, while liver tumors receive about 90 percent of their blood supply from arteries. This allows SIR-Spheres microspheres to target the liver tumors with a tumor-killing dose of radiation via the hepatic artery, while sparing surrounding healthy liver tissue. This typically isn’t possible with conventional external beam radiation.
How are SIR-Spheres microspheres different from other radiation treatments for cancers of the liver?
SIR-Spheres microspheres usually reduce liver tumors after just one treatment. The targeted nature of SIR-Spheres microspheres therapy enables doctors to deliver more radiation to the liver tumors than would be possible using conventional radiotherapy. Also, because traditional radiation is delivered to a general area of the body where cancer exists, it is less exact than SIR-Spheres microspheres. Another limitation of conventional radiotherapy is that it can only be applied to certain areas of the body, often not including the liver. SIR-Spheres microspheres, however, are designed specifically for delivery directly to the liver.
What are the goals of treatment using SIR-Spheres microspheres?
Clinical trials have shown SIRT increases the time-to-disease progression and overall survival without adversely affecting the patient’s quality of life.3 In clinical studies, SIR-Spheres microspheres have been combined with modern chemotherapy or administered as a monotherapy during a chemotherapy holiday and have been proven to: 4, 5-11
- Decrease the tumor burden in the liver 4, 5-9
- Increase time-to-disease progression 5, 6
- Increase survival time 3
- Potentially downsize tumors to liver resection or ablation 5, 7,10,11
- Provide palliation of symptoms
What side effects and complications can I expect?
When properly administered, most side effects are typically mild and subside within several days. Post treatment side effects can include abdominal pain and/or nausea which normally subside after a short time and/or with routine medication. Patients may also develop a mild fever that may last up to a week and fatigue which may last for several weeks. As a precaution, we may recommend additional medications with the aim of preventing or minimizing these side effects.
In rare instances there is a possibility that a small number of microspheres may inadvertently reach other organs in the body, such as the gall bladder, stomach, intestine or pancreas. If SIR-Spheres microspheres reach these organs, they may cause inflammation of the gall bladder (cholecystitis), stomach (gastritis) or intestine (duodenitis). These complications are rare, but if one of these occurs, they will require additional treatment. Your treatment team will have received special training to minimize these risks and to prevent them from happening.
SIR-Spheres microspheres are only for the treatment of colorectal cancer which has spread to the liver.12 For more information please contact us at info-us@sirtex.com.
*To see the full indication and important safety information visit http://www.sirtex.com/us/patients/about-sir-spheres-microspheres/important-risk-information/.
More Info:
Special Delivery – Dr. Dalsania on SIRT (DeSoto Times)
Local Health Alert: Y90 Cancer Treatment (Local Memphis)
More Memphis Area Liver Patients Receive Y-90 Treatment (Memphis Medical News)
SIR-Spheres® is a registered trademark of Sirtex SIR-Spheres Pty Ltd.
REFERENCES 1 Landis SH, Murray T, Bolden S, et al. Cancer Statistics. CA Cancer J Clin. 1999;49:8-31.
2 American Cancer Society. Cancer Facts & Figures, 2013. Atlanta, ACS, 2013: 5.
3 Seidensticker R, Denecke T, Kraus P, et al. Matched-pair comparison of radioembolization plus best supportive care versus best supportive care alone for chemotherapy refractory liver-dominant colorectal metastases. Cardiovasc Intervent Radiol. 2011 July 29. [Epub ahead of print]
4 Jakobs TF, Hoffman RT, Dehm K, et al. Hepatic yttrium-90 radioembolization of chemotherapy-refractory colorectal cancer liver metastases. J Vasc Interv Radiol. 2008;19(8):1187-1195.
5 Gray B, van Hazel G, Hope M, et al. Randomised trial of SIR-Spheres plus chemotherapy vs. chemotherapy alone for treating patients with liver metastases from primary large bowel cancer. Ann Oncol. 2001;12(12):1711–1720.
6 van Hazel G, Blackwell A, Anderson J, et al. Randomised phase 2 trial of SIR-Spheres plus fluorouracil/leucovorin chemotherapy versus fluorouracil/leucovorin chemotherapy alone in advanced colorectal cancer. J Surg Oncol. 2004;88(2):78–85.
7 Sharma R, van Hazel G, Morgan B, et al. Radioembolization of liver metastases from colorectal cancer using yttrium-90 microspheres with concomitant systemic oxaliplatin, fluorouracil, and leucovorin chemotherapy. J Clin Oncol. 2007;25(9):1099–1106.
8 van Hazel GA, Pavlakis N, Goldstein D, et al. Treatment of fluorouracil-refractory patients with liver metastases from colorectal cancer by using yttrium-90 resin microspheres plus concomitant systemic irinotecan chemotherapy. J Clin Oncol. 2009;27(25):4089–4095.
9 Kennedy A, Coldwell D, Nutting C, et al. Resin 90Y-microsphere brachytherapy for unresectable colorectal liver metastases: modern USA experience. International Journal of Radiation Oncology, Biology, and Physics. 2006;65(2):412-425.
10 Hoffman RT, Jakobs TF, Kubisch C, et al. Radiofrequency ablation after selective internal radiation therapy with Yttrium90 microspheres in metastatic liver disease—Is it feasible?. Eur J Radiol. 2010;74:199-205.
11 Whitney R, Tatum C, Hahl M, et al. Safety of hepatic resection in metastatic disease to the liver after yttrium-90 therapy. J Surg Res. 2011;166:236-240.
12 SIR-Spheres microspheres are indicated for the treatment of unresectable metastatic liver tumors from primary colorectal cancer with adjuvant intra-hepatic artery chemotherapy (IHAC) of FUDR (Floxuridine).